
苏州生物医药产业园1期A7-404
contact@togenx.com ; 138 1525 8535
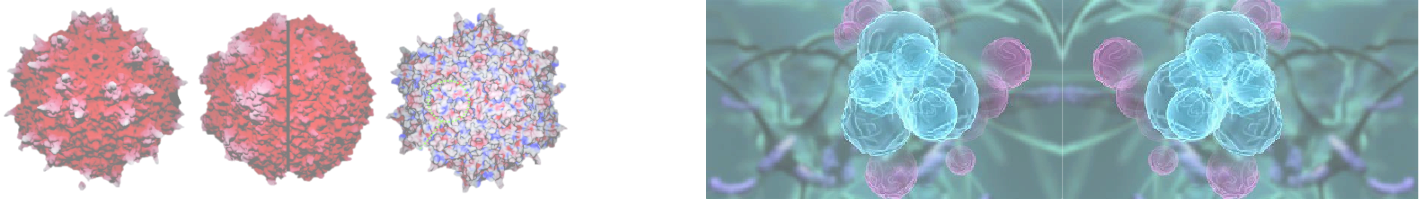
Adeno-associated virus:
A promising gene therapy vector
致力创新技术解决难题
推动基因疗法走向实用
ToGenx focuses on the innovation of key technologies for gene therapy, aiming to promote the rapid development of gene therapies and change the current situation of slow R&D relative to widespread market demand. In recent years, significant progress has been made in gene therapy, with 8 direct gene therapies available in the global market. Clearly, gene therapy is still in its early stages, urgently needing technological innovations to address numerous challenges, including viral vector manufacturing, tissue/cell targeting, and viral vector immunogenicity. These technical challenges have led to high costs for gene therapies, with a single treatment costing up to $4 million, severely limiting the practicality of gene therapies. ToGenx is advancing the industrialization of its innovative viral preparation technology system to solve the challenges of viral manufacturing for gene therapies. Ongoing innovations include the research and development of high-yield adeno-associated virus (AAV) technologies and the creation of cancer cell-specific AAV vectors. The creation of cancer cell-specific AAV vectors is an early step for ToGenx to pioneer a new approach to AAV cancer gene therapy.
@2x.png)
道基生物注重于基因疗法关键技术创新研发,以推动基因疗法快速发展,改变研发缓慢与广泛市场需求不相对称的现状。近年来基因疗法已经取得重大进展,全球市场已有 8个直接基因疗法。显然,基因疗法仍处在发展初期,急需技术创新来解决所面临的诸多挑战,包括病毒载体制备、组织细胞靶向、病毒载体免疫性等问题。技术上的挑战导致基因疗法价格高昂,一个疗程高达200-400万美元,严重限制了基因疗法的实用性。道基生物正在推动自主创新病毒制备技术的产业化,以解决基因疗法病毒制备难题。在研技术创新包括腺相关病毒 (AAV) 高产技术研发和癌细胞特异性AAV载体创建。创建癌细胞特异性AAV载体是道基生物开创AAV癌症基因疗法新途径的前期研发内容。
This is a completely new manufacturing technology for adeno-associated virus (AAV), which is primarily based on a new technological principle for effectively separating and purifying viral particles. It is simple, fast, high-quality, and efficient, making it very ideal. Patents have been granted in the USA, China, and the European Union. As planned, its industrialization includes pilot testing and a total of 3 phases of virus manufacturing industrial line assembly.
这是一种全新的新型腺相关病毒 (AAV) 制备技术,其核心在于采用新的技术原理有效的分离纯化病毒颗粒。简便快速、高质高效,非常理想。已获得美国、中国、及欧盟等专利授权。按计划,其产业化包括中试、及一共三期病毒制备工业生产线装配。
With the help of our self-developed virus preparation technology, we are currently conducting AAV capsid engineering, mainly focusing on creating cancer cell-specific AAV vectors, aimed at improving the specificity and effectiveness of treatments while reducing the dosage of viral vectors. This is part of ToGenx's preliminary efforts in new strategies for cancer gene therapy.
利用自主创新病毒制备技术优势,我们正在进行 AAV 病毒载体改造,以创建癌细胞特异性 AAV 载体为重点,旨在提高治疗的针对性和有效性、并减少病毒载体的用量。这是道基生物癌症基因疗法新战略的前期研发内容。
ToGenx (Suzhou) LLC Building A7,
Unit 404 (Phase 1) Suzhou Biomedicine
Industry Park Suzhou Industrial Park,
Suzhou Jiangsu Province, China
苏州生物医药产业园1期A7-404
江苏省苏州市苏州工业园区
@1x.png)
138 1525 8535
@1x.png)
info@togenx.com